Table of Content
- What is GMP Certification?
- Who Needs GMP Certification?
- What is the Difference Between GMP and ISO 22000?
- Key Principles of Quality Management in GMP Certification
- Key Requirements for GMP Certification?
- Why GMP Certification Matters?
- Process for Obtaining GMP Certification
- Common Challanges in Obtaining GMP Certification
What is GMP Certification?

GMP (Good Manufacturing Practice) certification is a regulatory quality assurance system that ensures products are consistently manufactured and controlled according to established safety and quality standards. It focuses on preventing contamination, mix-ups, errors, and defects during production through proper documentation, trained personnel, validated equipment, and controlled facilities.
GMP certification is widely applied in the pharmaceutical, food, cosmetic, nutraceutical, and medical device industries to protect consumer health and maintain regulatory compliance. GMP certification, along with ISO 13485, GDP, GHP, ISO 14001, and ISO 45001, strengthens operational discipline, traceability, and product reliability across manufacturing processes.
Who Needs GMP Certification?
GMP certification is essential for organisations involved in the manufacturing, processing, packing, or handling of regulated products to ensure consistent quality, safety, and compliance with industry standards.
GMP certification is required for:
- Pharmaceutical manufacturers
- Food and beverage manufacturers
- Nutraceutical and dietary supplement manufacturers
- Medical device manufacturers
- Cosmetic and personal care product manufacturers
- Herbal and Ayurvedic product manufacturers
- Contract manufacturing organisations (CMOs)
- Packaging and labelling units handling regulated products
What is the Difference between GMP and ISO 22000?
GMP and ISO 22000 differ in their focus, scope, regulatory requirements, and industry applicability, even though all aim to ensure quality and safety standards. Here are the key differences between GMP and ISO 22000:
|
Aspect |
GMP (Good Manufacturing Practice) |
ISO 22000 |
|
Focus |
Product safety and quality during manufacturing |
Food safety across the supply chain |
|
Issuing Authority |
National Regulatory Bodies |
|
|
Industry Applicability |
Pharmaceuticals, food, cosmetics, medical devices |
Food and beverage industry |
|
Objective |
Prevent contamination, mix-ups, and defects |
Control food safety hazards and ensure safe food |
|
Risk Management |
Focus on hygiene, contamination control, and batch integrity |
HACCP-based food safety risk management |
|
Standard Type |
Regulatory compliance standard |
International food safety standard |
What are the Key Principles of Quality Management in GMP Certification?

GMP certification is built on strong quality management principles that ensure products are consistently manufactured, controlled, and safe for use. Below are the key principles of quality management in GMP certification:
- Customer Focus: The organisation must focus on enhancing customers' experience and reliability of the product by ensuring it meets quality standards and regulatory compliance.
- Leadership and Accountability: Management must possess strong leadership and ensure accountability across all departments.
- Storage and Distribution Control: The organisation should ensure an effective storage and distribution control system to ensure products reach customers without damage or deterioration.
- Product Quality Review: Enhancing product quality by analysing batch data, product details, complaints and reviews to ensure the product consistently meets customers’ needs and requirements.
- Raw Material Control: Ensuring that the raw material is identified, documented, and quality-tested before use.
- Continuous Improvement: Striving for continuous product improvement through audits, performance reviews, and monitoring systems to enhance efficiency, minimise risks, and strengthen quality assurance practices over time.
- Change Control Management: Reviewing and monitoring changes or modifications in raw materials, equipment, processes, or suppliers to ensure that they do not affect the product quality, safety, or legal compliance.
- Hygiene and Sanitation Control: Adopting strict hygiene practices to prevent contamination, safeguard product integrity and ensure safe manufacturing conditions.
What are the Key Requirements for GMP Certification?

Here are the key-requirements for obtaining GMP certification:
1. Quality Management System: The manufacturer must plan and prepare a robust quality management system. It should include documented procedures, defined responsibilities, regular reviews, and corrective action systems to consistently meet regulatory and quality standards required for GMP certification.
2. Trained and Competent Personnel: All staff involved in production, quality control, and related functions must be trained in GMP principles. Regular training updates knowledge on hygiene, safety, and documentation practices, helping prevent errors and maintain consistent product quality.
3. Facility and Equipment Standards: Manufacturing premises and equipment must be designed, installed, and maintained to minimise contamination and ensure controlled production environments. Facilities should support clean operations and maintenance to uphold process reliability.
4. Documentation and Record Keeping: The manufacturer must prepare accurate, controlled records of manufacturing processes, batch information, quality checks, and equipment maintenance. Detailed documentation enables traceability, supports audits, and demonstrates consistent compliance with GMP requirements.
5. Quality Control and Testing: Management must implement an effective quality control and testing system to regulate raw material testing, product testing, and verify that products meet defined safety and quality standards before release into the market.
6. Handling and Storage of Materials: Raw materials, intermediates, and finished goods must be appropriately identified, handled, and stored to prevent mix-ups or contamination. This maintains integrity throughout the supply chain.
7. Complaint Handling and Product Recall Procedures: Manufacturers should establish effective mechanisms to record and investigate customer complaints. Manufacturers should also be prepared to quickly recall defective products to protect consumer safety and uphold compliance.
Why GMP Certification Matters?

GMP certification is crucial because it ensures manufacturing quality, protects consumer safety, and strengthens market trust for regulated products. Here is why GMP certification matters for manufacturers and retailers:
- Ensures Product Safety: Confirms that products are made under safe, controlled conditions to prevent defects or contamination.
- Builds Customer Confidence: Certification signals reliability and quality, increasing trust among buyers, patients, and partners.
- Regulatory Compliance: Demonstrates adherence to national and international manufacturing standards, reducing legal risks.
- Market Access & Growth: Many suppliers, retailers, and export markets demand GMP compliance as a prerequisite.
- Reduces Defects & Waste: Standardised systems improve consistency, decrease errors, and optimise resource use.
- Supports Brand Reputation: Shows commitment to quality, enhancing corporate credibility and competitive advantage.
What is the Process for Obtaining GMP Certification?
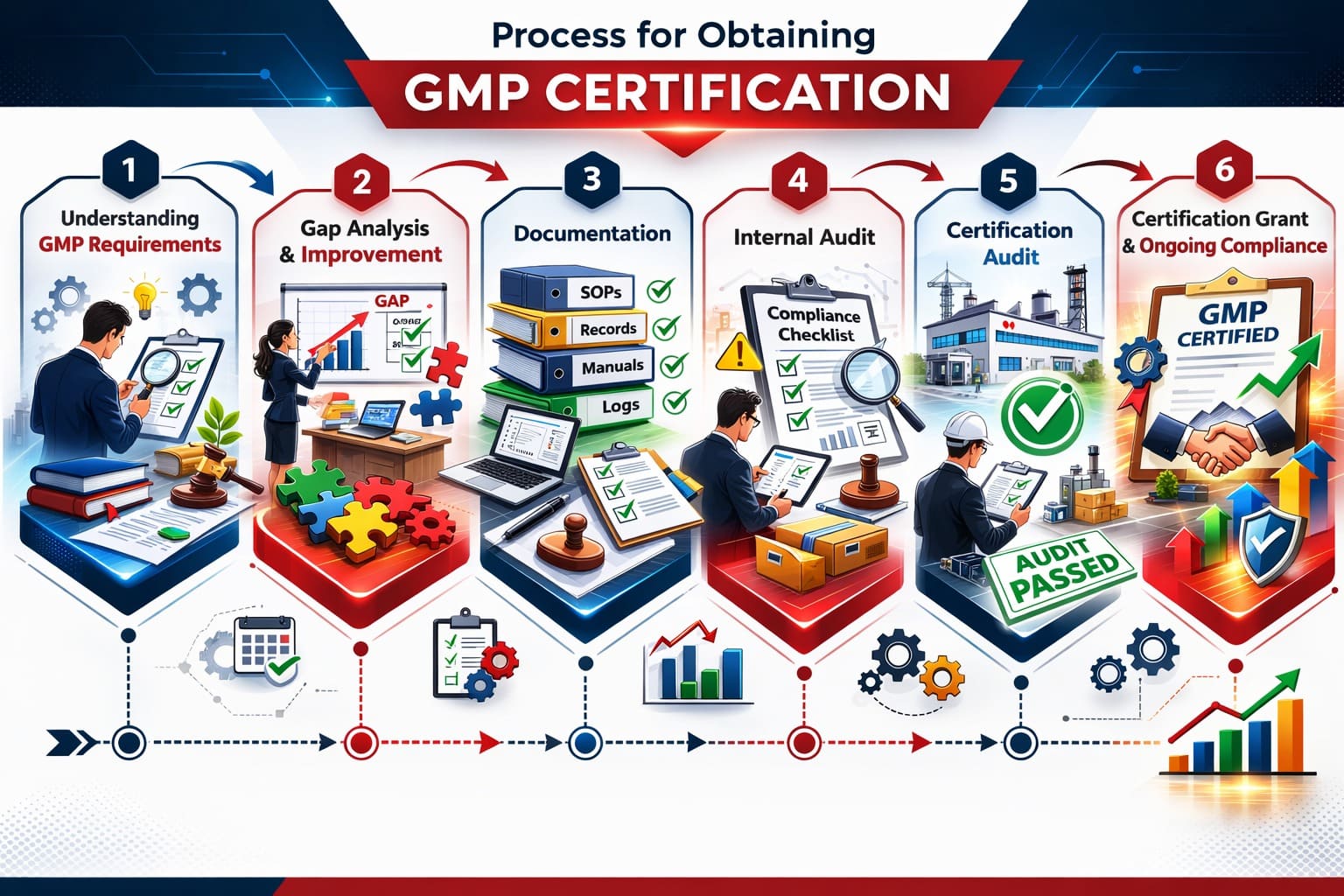
Here is the step-by-step process to obtain GMP certification hassle-free without any mistake:
1. Understanding GMP Requirements
The first step is to understand the GMP regulations applicable to your industry. A proper understanding ensures your organisation knows what changes are required before applying for certification.
2. Gap Analysis and Improvement
A gap analysis is a must to compare your current manufacturing practices with GMP requirements. This helps in identifying weaknesses in documentation, facilities, training, and quality systems. Based on the gap analysis findings, necessary improvements must be implemented.
3. Documentation
Prepare and update all required documents, such as SOPs, batch manufacturing records, validation reports, quality manuals, training records, and maintenance logs. Proper documentation demonstrates that processes are defined, controlled, and consistently followed across manufacturing operations.
4. Internal Audit
Before applying for certification, the manufacturers must perform an internal audit to assess compliance readiness. This helps identify non-conformities and operational weaknesses. Corrective and preventive actions should be implemented to ensure full compliance before the external audit.
5. Certification Audit
An accredited certification body conducts an on-site audit to evaluate facilities, processes, equipment, and documentation. Auditors verify that GMP standards are effectively implemented and consistently followed in manufacturing processes.
6. Certification Grant and Ongoing Compliance
If compliance is successfully demonstrated, GMP certification is granted. However, maintaining certification requires continuous monitoring, internal audits, updated documentation, and adherence to regulatory changes to ensure long-term compliance.
What are the Common Challanges in Obtaining GMP Certification?
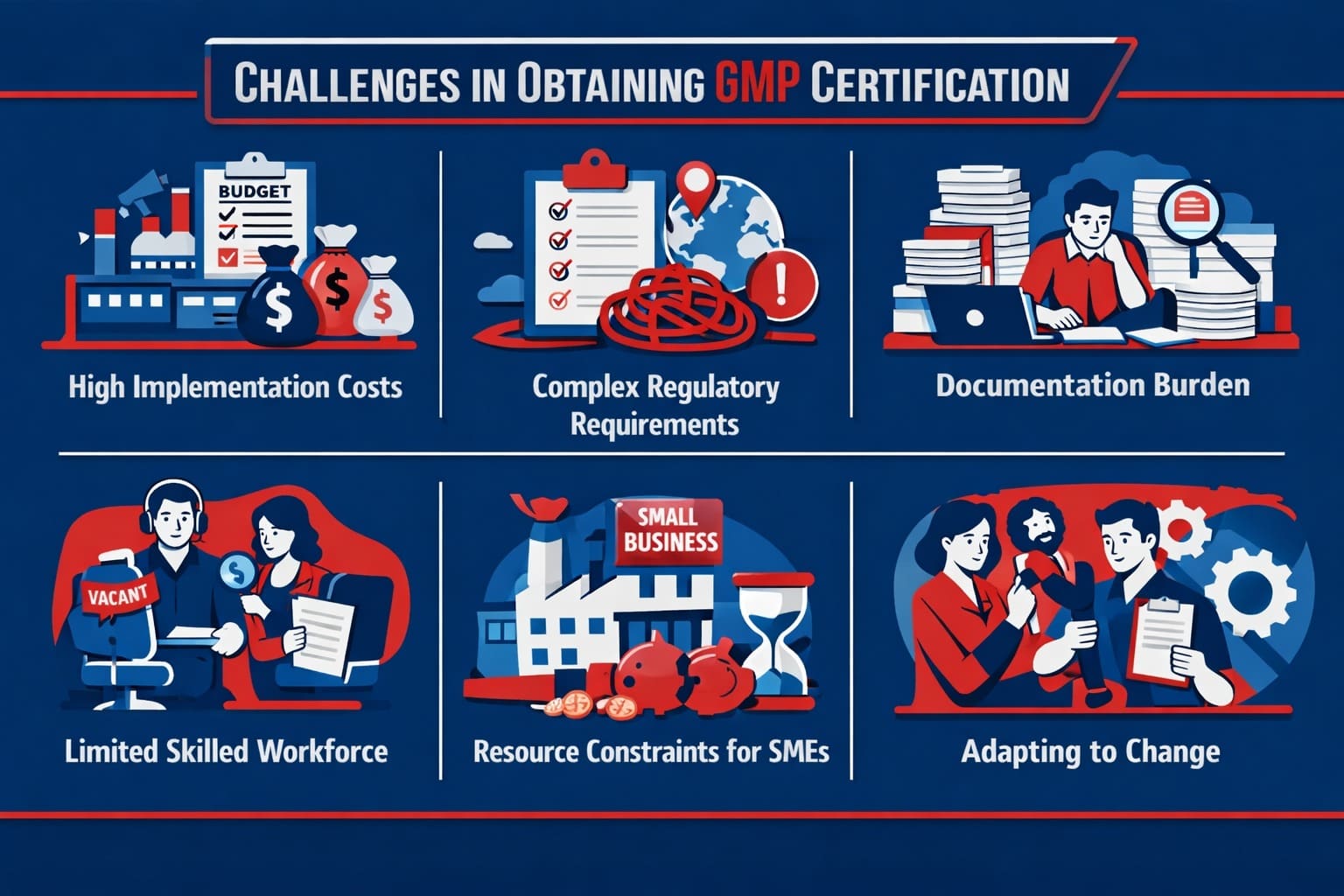
Obtaining GMP certification can be challenging due to regulatory complexity, extensive documentation and legal rules. Here are the common challenges manufacturers can face during the GMP certification process:
- High Implementation Costs: Setting up compliant facilities and quality systems requires substantial investment in infrastructure, equipment, and training.
- Complex Regulatory Requirements: Navigating detailed and evolving GMP standards, often differing across regions, can be overwhelming.
- Documentation Burden: Maintaining accurate and extensive records for processes, validation, and audits is resource-intensive.
- Limited Skilled Workforce: Finding and retaining personnel experienced in GMP practices and quality assurance can be difficult.
- Resource Constraints for SMEs: Smaller companies often struggle with budget, manpower, and infrastructure limitations.
- Adapting to Change: Implementing and managing changes in processes and systems for compliance can face resistance and logistical challenges.
Get Your GDP Certification with Expert Guidance Today!
GDP certification ensures safe storage, handling, and transportation of medicinal products while strengthening regulatory compliance and supply chain integrity. By implementing structured quality systems and proper documentation, businesses can reduce risks and enhance market credibility. You can easily apply for GDP certification anytime with us to streamline compliance, pass audits confidently, and build a reliable, globally trusted pharmaceutical distribution network.
FAQS
Have some questions about GMP Certification?
We have got you covered:
-
Why is GMP certification important for manufacturers?
GMP certification ensures consistent manufacturing standards that protect consumers and maintain regulatory compliance.
- How does GMP certification protect consumers?
- What role does management play in GMP certification?
- How does GMP certification support export opportunities?
- How often are audits conducted under GMP certification?
- Does GMP certification apply to packaging units?
- How does GMP certification improve operational efficiency?
- Is facility design important for GMP certification?
- Does GMP certification require equipment validation?
- How does GMP certification manage production deviations?
- Can herbal product manufacturers obtain GMP certification?
- Does GMP certification require supplier evaluation?
- Is it compulsory for manufacturers to obtain GMP certification?
- What is the typical timeline required to complete GMP certification?
- For how many years does GMP certification remain valid?
- Can a newly established company secure GMP certification?
- What factors influence the cost of GMP certification?
- What happens if non-conformities are identified during a GMP certification audit?
- Is GMP certification recognised across different countries?
- Can GMP certification be suspended after being granted?
