Table of Content
- What is GHP Certification?
- Which Industries Benefit from GHP Certification?
- Difference Between GHP and GMP
- Key authorities that Regulates GHP Certification in India
- Key Principles of Quality Management in GHP Certification
- Key Requirements for GHP Certification
- Benefits of GHP Certification
- Validity Period for GHP Certification?
- Process for Obtaining GHP Certification
- The Bottom Line!
What is GHP Certification?

GHP (Good Hygiene Practice) Certification is a quality standard that focuses on ensuring a high level of cleanliness, hygiene, and disinfection in the manufacturing and processing environment. This certification demonstrates that the products, particularly food, beverages, cosmetics, and pharmaceuticals, are processed, packed, and produced in a safe and hygienic manner.
GHP certification ensures adherence to HACCP principles, FSSC 22000 requirements, and adherence to best hygienic practices. It guarantees that the product being processed is suitable for human consumption and complies with all regulatory rules and requirements.
Who Needs GHP Certification?
GHP certification is important for industries within the food supply chain and other sectors handling consumer health and safety products. The industries that benefits from GHP certification includes:
- Food and Beverage Manufacturing
- Hospitality and Catering
- Retail and Supermarkets
- Logistics and Warehousing
- Pharmaceuticals and Cosmetics
- Agriculture
What is the Difference Between GHP and GMP?
The GHP certification deals with good and safe hygienic/cleanliness practices, while the GMP certification deals with controlling and managing manufacturing processes. Here is the key difference between GHP (Good Hygiene Practices) and GMP (Good Manufacturing Practices):
|
Basis of Difference |
GHP (Good Hygiene Practices) |
GMP (Good Manufacturing Practices) |
|
Primary Focus |
Focuses on maintaining hygiene and sanitation in food handling and processing environments |
Focuses on controlling manufacturing processes to ensure consistent product quality and safety |
|
Objective |
Prevents contamination from biological, chemical, and physical hazards |
Ensures products are consistently produced and controlled according to quality standards |
|
Scope |
Covers cleaning, sanitation, pest control, personal hygiene, and facility maintenance |
Covers production processes, quality control, documentation, equipment validation, and personnel training |
|
Nature of Standard |
Basic prerequisite hygiene program |
Comprehensive quality assurance system |
What are the Key Authorities that Regulate GHP in India?
In India, GHP certification is regulated by central food safety authorities and accreditation bodies that ensure food businesses comply with hygiene, sanitation, and public health standards. Here are the key authorities that govern GHP certification in India:
|
Authority |
Establishment Year |
Details |
|
2008 |
Apex regulatory authority responsible for framing and enforcing food safety and hygiene standards in India. It ensures implementation of GHP requirements through inspections, licensing, and compliance monitoring. |
|
|
1976 |
The central ministry that oversees FSSAI and formulates national policies related to public health, food safety, and hygiene regulations. |
|
|
National Accreditation Board for Certification Bodies (NABCB) |
1996 |
Provides accreditation to certification bodies that issue GHP and food safety certifications, ensuring credibility and compliance with international standards. |
What are the Key Principles of Quality Management in GHP Certification?

Here are the key principles of quality management in GHP certification:
- Facility Hygiene Control: Ensuring the overall facility is disinfected, well-ventilated, and hygienic.
- Cleaning and Sanitation Programmes: Emphasising cleaning and sanitation programmes in the facility to prevent contamination risk and ensure the safety, efficacy, and quality of products.
- Risk Identification and Control: Recognising biological, chemical, and physical hazards and taking suitable control or preventive measures.
- Systematic Hygiene Monitoring: Emphasising regular monitoring and review of hygienic practices to detect issues early and resolve them timely, supporting discipline and product safety.
- Process Approach: Establishing a process-orientated system to ensure every stage from raw material procurement to finished product followed defined hygienic protocols and procedures.
- Continuous Hygiene Improvement: Focusing on ongoing processes, procedures, performance to evaluate the current position and taking suitable steps for continuous improvement and long-term compliance.
What are the Key Requirements for GHP Certification?

GHP certification requires establishing mandatory hygiene and sanitation practices to ensure safe production, handling, and storage of products. Here are the key requirements of GHP certification:
1. Cleaning and Disinfection Control: The organisation must prepare and implement a well-documented cleaning and disinfection programme. It should cover equipment cleaning, define methods and procedures, and specify effective contamination control measures. This reduces chemical infusion and ensures a safe production environment.
2. Personal Hygiene and Health Control: The employees must follow a strict and regulated occupational health and safety programme, including proper handwashing, hair restraints, illness reporting, and protective clothing. This prevents contamination and ensures compliance with safety standards.
3. Pest Control Management System: Preparing an effective pest control management system is a must in GHP certification. The organisation must regularly update the pest control system, monitor performance, and maintain good waste management practices.
4. Water and Utility Safety: The organisation must maintain pipelines, water treatment systems, storage tanks, and compressed air in the facility to prevent chemical hazards or microbial hazards.
5. Raw Material and Storage Controls: GHP certification requires a robust raw material management system, including material tracing, supplier management, and quality management. It also emphasises proper storage facilities to prevent spoilage, contamination or deterioration of the raw material.
6. Cross-Contamination Prevention Measures: GHP certification requires the organisation to maintain physical separation of raw material from finished products. It requires using colour-coded equipment, dedicated tools, and defined roles and responsibilities to prevent mixing or contamination of products.
7. Documentation and Record Maintenance: The organisation must maintain detailed records of cleaning schedules, pest control management systems, raw material procurement, suppliers, temperature monitoring reports, and risk management practices to facilitate audits and certification application or approval.
What are the Benefits of GHP Certification?
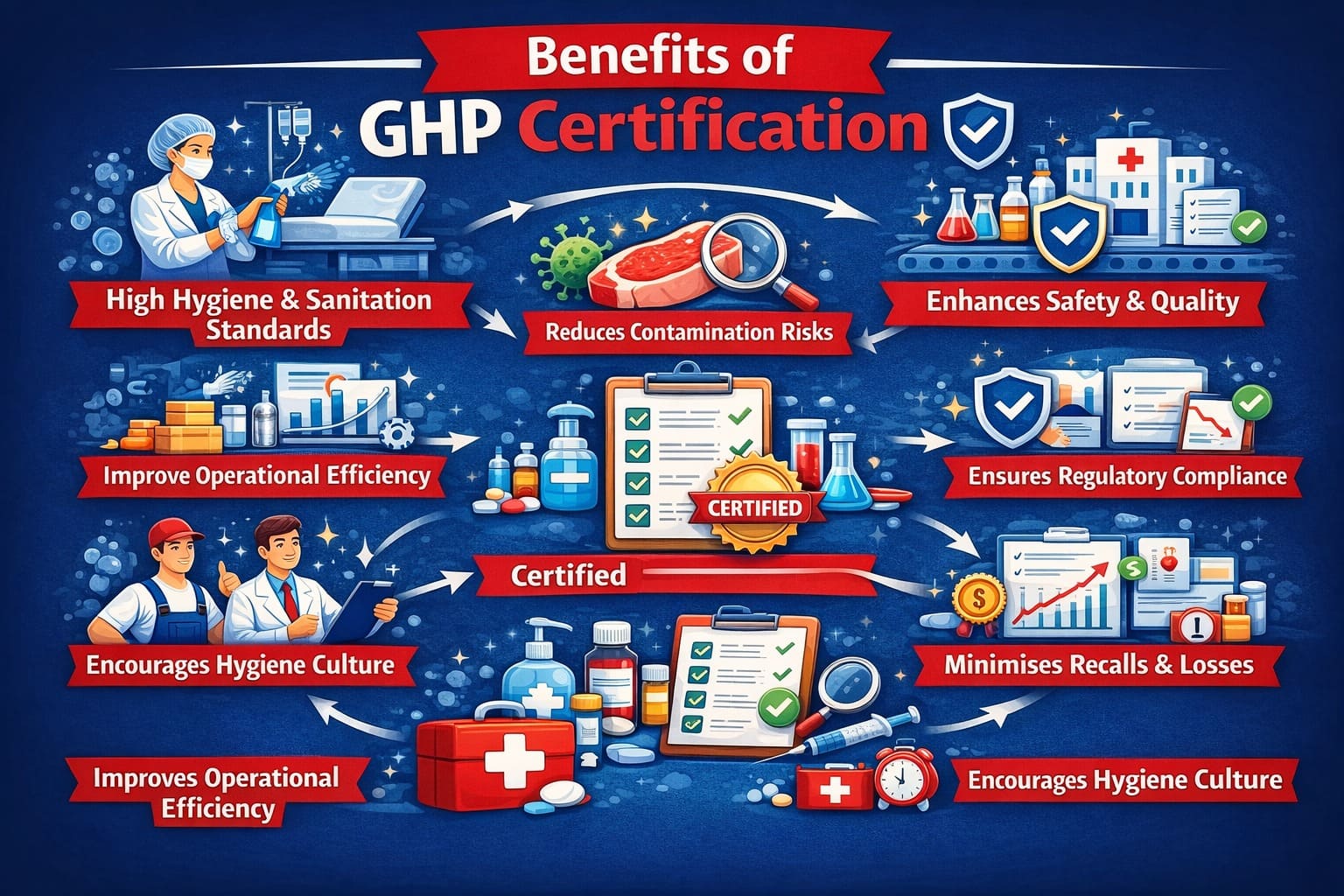
Here are the key benefits of GHP certification that make it important and a must-have for manufacturers and retailers:
- Ensures high standards of hygiene and sanitation practices.
- Reduces risk of contamination and food safety hazards.
- Enhances product safety, quality, and consumer confidence.
- Improves operational efficiency through structured hygiene procedures.
- Supports compliance with national and international regulatory requirements.
- Strengthens brand reputation and market credibility.
- Minimises product recalls, complaints, and financial losses.
- Encourages employee awareness and a hygienic work culture.
- Facilitates easier audits and regulatory inspections.
- Promotes continuous improvement in hygiene management systems.
What is the Validity Period for GHP Certification?
The GHP certification is usually valid for 3 years, subject to the successful completion of annual audits or surveillance audits. If any non-conformity is found during the annual audit, the certification may be suspended or cancelled before the period of three years.
Moreover, after 3 years, the organisation needs to renew the certification by demonstrating effective or continuous compliance with all the requirements of the certification.
What is the Process for Obtaining GHP Certification?

Here is the step-by-step process to easily obtain GHP certification:
1. Application Submission
The organisation begins by submitting a formal application to the certification body. This includes business details, scope of activities, product categories, facility information, and required documentation.
2. Documentation Review
In this stage, the organisation prepares and updates all the documentation, including hygiene policies, procedures, SOPs, cleaning schedules, pest control records, and compliance reports. The purpose is to ensure the organisation aligns with all the GHP requirements.
3. Internal Audit and Management Review
Before the external audit, the organisation conducts an internal audit to assess compliance with GHP standards. Top management reviews audit findings, corrective actions, and system performance. This ensures gaps are identified and resolved internally, demonstrating readiness for the certification audit.
4. Certification Audit and On-Site Inspection
The authorised certification body visits the facility to verify practical implementation of hygiene and sanitation practices. They inspect cleaning processes, employee hygiene, storage systems, pest control measures, and documentation records. Any non-conformities are recorded, and corrective actions are required before certification approval.
5. Certification Decision
After reviewing audit findings and corrective actions, the certification body makes a final decision. If all requirements are met, the GHP certificate is issued. If major non-conformities remain unresolved, certification may be delayed until compliance is demonstrated.
6. Surveillance Audit
The certification body conducts periodic surveillance audits to ensure continuous compliance with GHP standards. These audits verify that hygiene systems are consistently maintained and improved.
What are the Key Challenges in Obtaining GHP Certification?

Obtaining GHP certification can be challenging for businesses that lack structured hygiene systems, proper documentation, and trained personnel. Many food operators struggle with implementation gaps, regulatory understanding, and audit preparedness, which can delay certification approval and increase compliance risks. Here are some challenges that businesses usually face:
- Inadequate Documentation: Missing or poorly maintained SOPs, cleaning schedules, and hygiene records.
- Lack of Employee Training: Staff unaware of personal hygiene standards and sanitation protocols.
- Infrastructure Gaps: Improper facility layout leading to cross-contamination risks.
- Poor Pest Control Systems: Ineffective monitoring and absence of preventive pest management programs.
- Weak Internal Audits: Failure to identify non-conformities before the certification audit.
- Regulatory Misunderstanding: Limited awareness of FSSAI hygiene requirements and compliance standards.
The Bottom Line!
GHP certification plays a vital role in ensuring consistent hygiene, sanitation, and food safety practices across manufacturing, processing, and retail operations. It not only helps organisations comply with regulatory requirements but also strengthens consumer trust and brand credibility. By implementing structured hygiene controls and continuous monitoring systems, businesses can reduce contamination risks and improve operational efficiency. To successfully obtain your GHP certification hassle-free and timely, you can contact us!
FAQS
Have some questions about GHP certification?
We have got you covered:
-
How does GHP certification improve food safety?
It enforces structured hygiene controls that prevent microbial, chemical, and physical contamination risks.
- Is GHP certification mandatory in India?
- How much time does it take to obtain GHP certification?
- What is the cost of GHP certification?
- What is the role of internal audit in GHP certification?
- What happens during a GHP certification audit?
- Can small businesses apply for GHP certification?
- How does GHP certification reduce contamination risks?
- How does GHP certification support regulatory compliance?
- What is the difference between GHP and HACCP certification?
- Is training required for GHP certification?
- Can GHP certification help during inspections?
- How often are surveillance audits conducted under GHP certification?
- Can GHP certification be suspended?
- Does GHP certification improve brand reputation?
- What role does management play in GHP certification?
- How does GHP certification improve operational efficiency?
- Can GHP certification prevent product recalls?
- Is documentation mandatory for GHP certification?
- What is the scope of GHP certification?
